One of the most active fields of clinical investigation in immuno-oncology today is augmenting responses to checkpoint [CTLA4 or PD-(L)1] inhibition therapy by re-sensitizing tumors that were initially unresponsive or had stopped responding to treatment. Continue reading
Tag Archives: PD-L1
Adenosine – a critical checkpoint in the tumor microenvironment
The tumor microenvironment (TME) includes a host of cells (mesenchymal, immune, vascular), cytokines, and other signaling molecules that serve to abrogate the innate and adaptive immune responses against the tumor. This is appropriate to maintain tissue homeostasis, and to prevent autoimmunity after the rogue cancer cells have been eliminated. Cancer cells co-opt many of these pathways to terminate the effective immune response so that they are not wiped out, rather, proliferate, invade, metastasize and kill, Continue reading
Priming cancer for immunotherapy
Augmenting the responses to checkpoint inhibitors, which remove the “breaks” from the immune response, is a very popular area of research. The general concept is to turn immunologically cold tumors hot. For example, triple negative breast cancer (TNBC) is considered an immunologically cold tumor – anti-PD(L)1 therapy has shown responses of just 5-10%. Continue reading
Olaparib – PARP inhibitor for triple negative breast cancer
Olaparib (Lynparza) is a PARP (poly-ADP ribose polymerase) inhibitor that was approved by the FDA in 2014 for the treatment of patients with advanced ovarian cancer who have mutated BRCA1,2 gene. Recently, the drug showed a 70% reduction in risk of progression in patients with less-advanced disease in the maintenance therapy setting:
The Phase III SOLO-2 trial demonstrated a significant improvement in progression-free survival (PFS) in germline BRCA-mutated (gBRCA), platinum-sensitive, relapsed ovarian cancer patients treated with Lynparza (olaparib) tablets (300mg twice daily) compared with placebo in the maintenance setting. The trial met its primary endpoint of investigator assessed PFS (HR 0.30; 95% CI 0.22 to 0.41; P<0.0001; median 19.1 months vs 5.5 months).
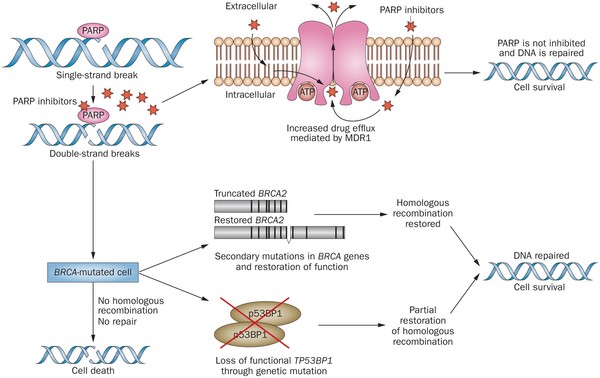
PARP inhibitors act in a counter-intuitive manner – by blocking PARP in the context of mutated BRCA1, the cell becomes overwhelmed with double strand breaks, leading to crisis and cell death. BRCA1 mutations, alone, predispose the cell to the accumulation of mutations in protooncogenes and tumor suppressor genes – a few double strand breaks are tumorigenic, whereas a massive number of double strand breaks, as occurs in the context of PARP inhibition, leads to apoptosis.
The use of PARP inhibitors for breast cancer makes great sense, However, in a Phase 3 trial of velparib, an experimental PARP inhibitor, failed to achieve better rates of complete pathogenic response in patients with triple negative breast cancer (TNBC – lack of HER-2, estrogen, and progesterone receptor up-regulation) versus chemotherapy, alone.
At the ASCO conference last week, AstraZeneca presented data on the use of olaparib in 302 patients with BRCA1,2 mutated breast cancer from its OlympiAD trial that compares olaparib against physician’s choice of chemotherapy (capecitabine 2500 mg/m2 d1-14 q 21, or vinorelbine 30 mg/m2 d1,8 q 21, or eribulin 1.4 mg/m2 d1,8 q 21):
OlympiAD Inclusion Criteria:
- Germline mutation in BRCA1 or BRCA2 that is predicted to be deleterious or suspected deleterious.
- Histologically or cytologically confirmed breast cancer with evidence of metastatic disease.
- Prior therapy with an anthracycline and a taxane in either an adjuvant or metastatic setting.
- Prior platinum allowed as long as no breast cancer progression occurred on treatment or if given in adjuvant/neoadjuvant setting at least 12 months from last dose to study entry elapsed.
- ER/PR breast cancer positive patients must have received and progressed on at least one endocrine therapy (adjuvant or metastatic), or have disease that the treating physician believes to be inappropriate for endocrine therapy.
- ECOG performance status 0-1.
- Adequate bone marrow, kidney and liver function.
OlympiAD Exclusion Criteria:
- Prior treatment with PARP inhibitor.
- Patients with HER2 positive disease.
- More than 2 prior lines of chemotherapy for metastatic breast cancer.
- Untreated and/or uncontrolled brain metastases.
Results were quite impressive – this was the first study that demonstrated PARP inhibition is effective in breast cancer:
- About 60% of patients saw their tumors shrink, a hair more than double the 29% objective response rate seen in those patients on chemotherapy.
- Lynparza showed efficacy in patients with TNBC, which is more difficult to treat. AbbVie, which is developing its own PARP inhibitor called veliparib, recentlyannounced a study specifically geared to look at veliparib’s activity in triple negative breast cancer failed to show a benefit when added to chemo.
- Additionally, treatment with Lynparza improved the time to second progression or death compared to chemo,suggesting patients who relapsed after Lynparza experienced a less aggressive return of their cancers.
Astrazeneca is studying olaparib with many combinations, including a study in TNBC with PD-L1 inhibitor durvalumab and CTLA-4 inhibitor tremelimumab.
Imfinzi, the latest approved checkpoint, and checkpoint combinations
The latest checkpoint inhibitor to be approved is AstraZeneca’s Imfinzi (durvalumab), a monoclonal antibody directed against PD-L1, which is expressed on cancer cells.
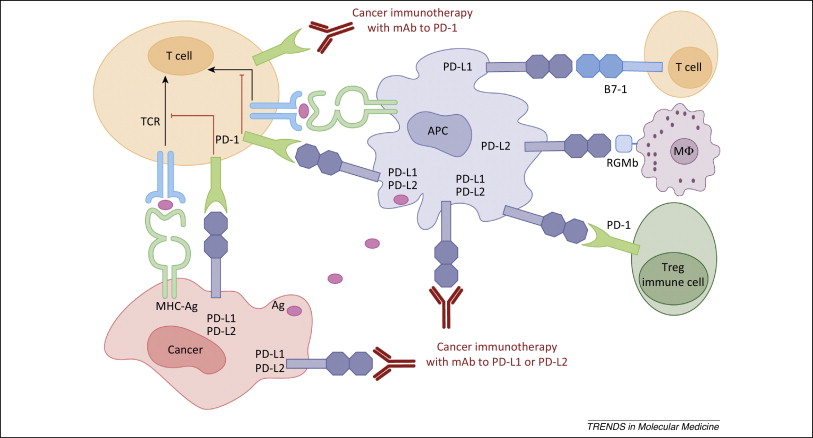
Figure 1. PD-1 / PD-L1 axis. https://www.cell.com/trends/molecular-medicine/references/S1471-4914(14)00183-X
Platelet conjugates effectively deliver checkpoint inhibitors to tumors
Platelets are the second most abundant cellular component of blood. The platelet membrane contains an abundance of receptors to facilitate interactions with subendothelial matrix , other blood cells, and other platelets. The central role of platelets is in hemostasis, however, they also contain copious amounts of cytokines that induce inflammation. Continue reading
PD-L1 Inhibitor, avelumab, approved for Merkel cell carcinoma
Avelumab (Bavencio) is a PD-L1 inhibitor that was approved for the treatment of patients with metastatic Merkel cell carcinoma (MCC). Continue reading
BMS and PsoOxus collaborate on transgenic oncolytic virus plus nivolumab
BMS paid PsiOxus $50MM upfront for exclusive rights to develop PsiOxus’ NG-348 enadenotucirev, a systemically administered oncolytic adenovirus therapeutic, in combination with Bristol-Myers Squibb’s Immuno-Oncology checkpoint inhibitor Opdivo (nivolumab) to treat a range of solid tumor types in late-stage cancer patients. This is a “big deal” – PsiOxus could receive up to $886 million in development, regulatory, and sales-based milestones, plus sales royalties. Continue reading
How PD-1 abrogates the anti-tumor immune response
PD-1 inhibition (Figure 1) has quickly become a front-line therapy for non-small cell lung cancer and melanoma. Moreover, PD-1 and PD-L1 inhibitors are being tested in combination with other checkpoint inhibitors, targeted therapies, cancer vaccines, monoclonal antibodies, and other modalities. But, how does PD-1 blunt the anti-tumor immune response? Continue reading
Celgene Collaborates with Jounce Therapeutics on ICOS T-cell Stimulator
Checkpoint inhibitors, alone, are effective in 25% of patients when administered as a single agent. The goal of the collaboration between Celgene and Jounce is to address the other 75% of patients. The lead program is focused on ICOS, the Inducible T cell CO-Stimulator, a protein on the surface of T cells that can spur an immune response against a patient’s cancer. Continue reading