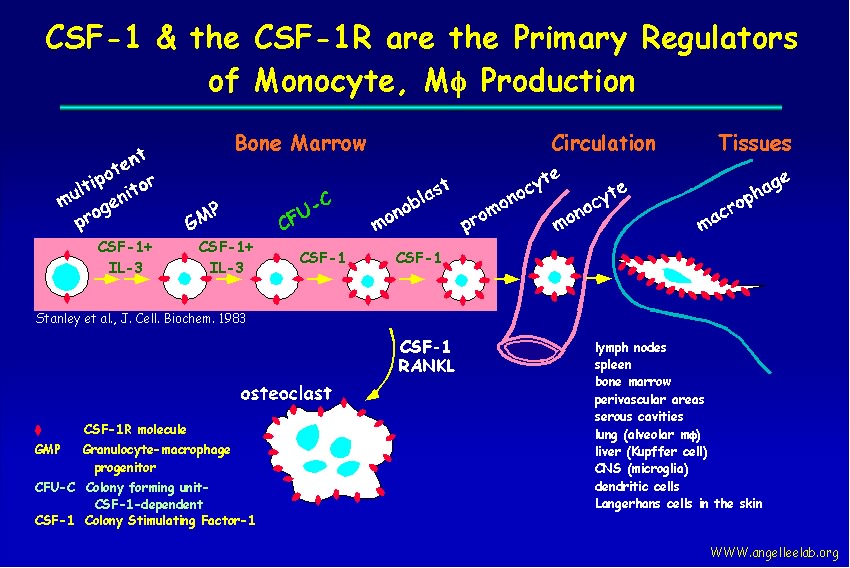
Colony Stimulating Factor (CSF-1) is an essential growth factor for cells of the monocyte-macrophage lineage, including osteoclasts.
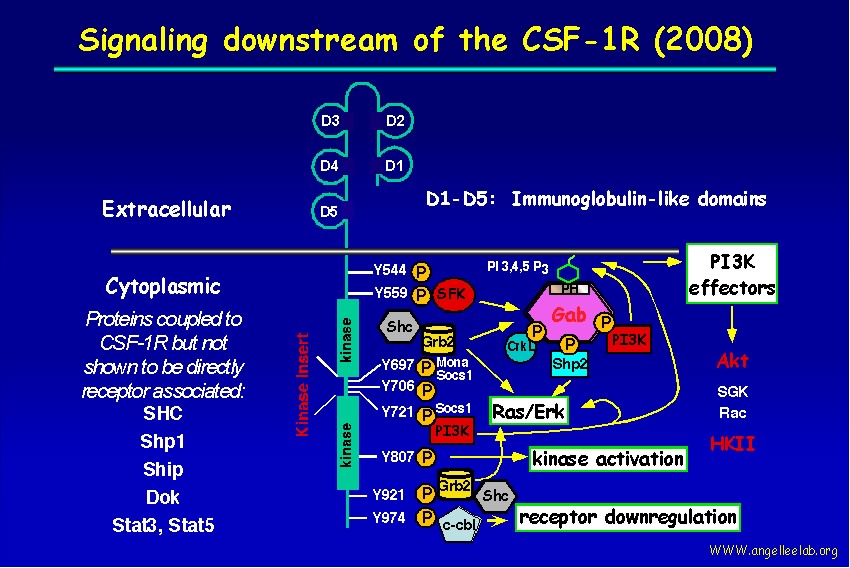
 CSF-1R is a receptor tyrosine kinase that activates many signaling pathways responsible for growth.
CSF-1R is a receptor tyrosine kinase that activates many signaling pathways responsible for growth.
Colony stimulating factor 1 (CSF-1) also recruits tumor-infiltrating myeloid cells (TIM) that suppress tumor immunity, including M2 macrophages and myeloid-derived suppressor cells (MDSC). Tumor-associated macrophages (TAMs) interact with myeloid-derived suppressor cells (MDSCs) and T cells in the tumor microenvironment, the end result of which is to promote a Th2-type polarized environment with high levels of CD4+ T cells, and low levels of CD8+ cytotoxic T cells, which promotes tumor growth.
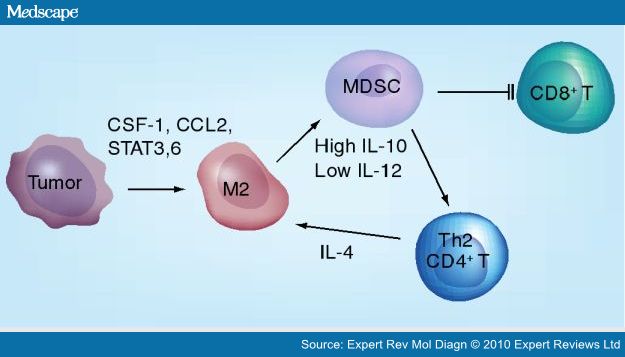
Certain breast cancers produce CSF-1, CCL2, STAT3 and STAT6, which promote macrophage infiltration and M2 differentiation. High Th2 CD4+ T cells with low CD8+ T cells results in a protumoral environment with increased metastatic risk. Interactions between M2 macrophages and MDSCs lead to high levels of IL-10 and low levels of IL-12, further reinforcing the M2 phenotype and increasing levels of Th2-type CD4+ T cells. These CD4+ T cells produce IL-4, which also polarizes macrophages toward M2, creating a feedback loop. Meanwhile, CD8+ T cells are suppressed, resulting in an overall immune-permissive environment for tumor growth and spread. CSF: Colony-stimulating factor; MDSC: Myeloid-derived suppressor cell.
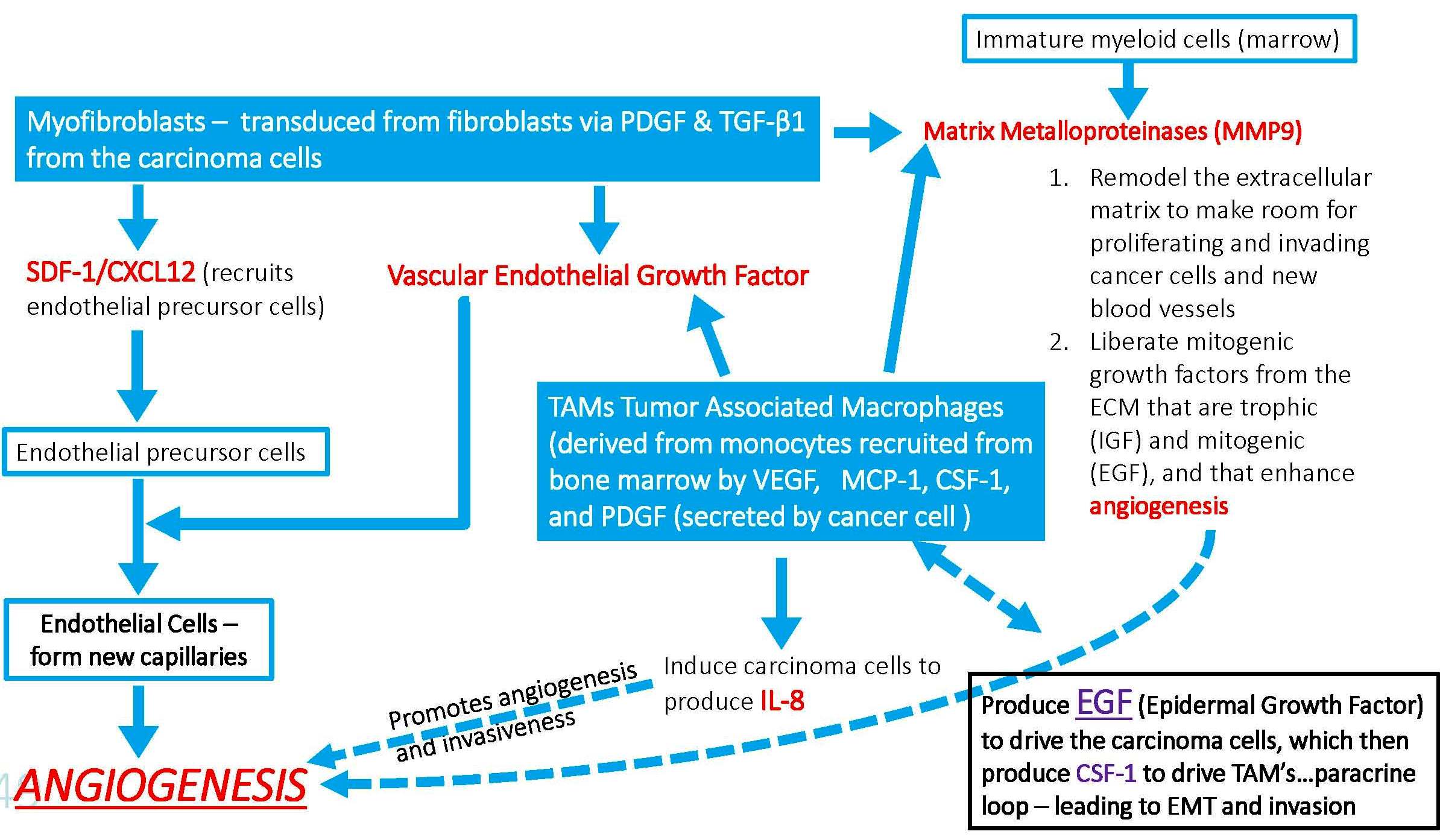
In cancer, aberrant expression of CSF-1 and CSF-1R has been found in cancers of the breast, ovaries and endometrium, correlating with disease progression and malignancy. CSF-1 is a biomarker for postmenopausal breast cancer risk. A role for CSF-1-dependent macrophages in breast cancer is well supported: 1) strong correlation between poor prognosis and TAMs (tumor associated macrophages); 2) CSF-1 is important for the angiogenic switch and progression to malignancy through recruitment and activation of tumor-associated macrophages, which secrete matrix metalloproteinases and VEGF, and induce cancer cells to produce IL-8.
Plexxikon is developing PLX3397 for the treatment of breast cancer, pigmented villonodular synovitis or giant cell tumor of the tendon sheath, prostate cancer, acral (palms and soles) and mucosal melanoma, neurofibroma, glioblastoma, and acute myeloid leukemia.
Merck entered into a collaboration with Plexxikon to combine PLX3397 with its PD-1 (immune checkpoint injibitor) antibody, Keytruda (pembrolizumab). The rationale for combining CSF-1 and Keytruda include:
- Multiple mechanisms of action: CSF-1 inhibition blocks invasion and metastases of cancer cells, while Keytruda prevents the abrogation of the immune attack on cancer cells;
- Complementary actions in heterotypic cellular interactions in the cancer micro-environment: CSF-1 inhibits the function of MDSCs, a subset of heterogeneous bone marrow-derived hematopoietic cells that are found in the peripheral blood of cancer patients and positively correlate to malignancy. Solid tumors contain MDSCs that maintain an immune-suppressive network in the tumor microenvironment. So, Keytruda and CSF-1 may act in a complementary fashion in restoring cancer immunity.