Janssen Biotech acquired a worldwide license to Alligator Bioscience’s Phase I immunotherapy ADC-1013, an agonistic monoclonal antibody that targets CD40, an immuno-stimulatory receptor found on antigen-presenting cells, which can spur an increase in T cells attacking a tumor.
CD40 is expressed on antigen presenting cells. Activated T-cells up-regulate CD40 ligand, which then engage cognate receptors on APCs.
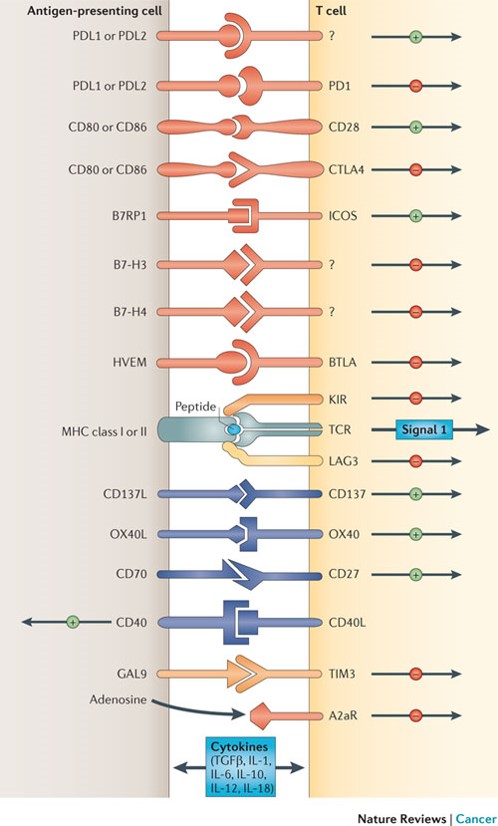
Figure 1: Depicted are various ligand–receptor interactions between T cells and antigen-presenting cells (APCs) that regulate the T cell response to antigen (which is mediated by peptide–major histocompatibility complex (MHC) molecule complexes that are recognized by the T cell receptor (TCR)). These responses can occur at the initiation of T cell responses in lymph nodes (where the major APCs are dendritic cells) or in peripheral tissues or tumours (where effector responses are regulated). In general, T cells do not respond to these ligand–receptor interactions unless they first recognize their cognate antigen through the TCR. Many of the ligands bind to multiple receptors, some of which deliver co-stimulatory signals and others deliver inhibitory signals. In general, pairs of co-stimulatory–inhibitory receptors that bind the same ligand or ligands — such as CD28 and cytotoxic T-lymphocyte-associated antigen 4 (CTLA4) — display distinct kinetics of expression with the co-stimulatory receptor expressed on naive and resting T cells, but the inhibitory receptor is commonly upregulated after T cell activation. One important family of membrane-bound ligands that bind both co-stimulatory and inhibitory receptors is the B7 family. All of the B7 family members and their known ligands belong to the immunoglobulin superfamily. Many of the receptors for more recently identified B7 family members have not yet been identified. Tumour necrosis factor (TNF) family members that bind to cognate TNF receptor family molecules represent a second family of regulatory ligand–receptor pairs. These receptors predominantly deliver co-stimulatory signals when engaged by their cognate ligands. Another major category of signals that regulate the activation of T cells comes from soluble cytokines in the microenviron-ment. Communication between T cells and APCs is bidirectional. In some cases, this occurs when ligands themselves signal to the APC. In other cases, activated T cells upregulate ligands, such as CD40L, that engage cognate receptors on APCs. A2aR, adenosine A2a receptor; B7RP1, B7-related protein 1; BTLA, B and T lymphocyte attenuator; GAL9, galectin 9; HVEM, herpesvirus entry mediator; ICOS, inducible T cell co-stimulator; IL, interleukin; KIR, killer cell immunoglobulin-like receptor; LAG3, lymphocyte activation gene 3; PD1, programmed cell death protein 1; PDL, PD1 ligand; TGFβ, transforming growth factor-β; TIM3, T cell membrane protein 3. https://www.nature.com/nrc/journal/v12/n4/fig_tab/nrc3239_F1.html
What does ADC 1013 do?
The functional activity of ADC-1013 was investigated in human and murine in vitro models. The in vivo effect was investigated in two separate bladder cancer models, both using human xenograft tumors in immune deficient NSG mice and using a syngeneic bladder cancer model in a novel human CD40 transgenic mouse.
Activation of dendritic cells (DC) by ADC-1013 results in upregulation of the costimulatory molecules CD80 and CD86, and secretion of IL12. ADC-1013 also activates DCs from human CD40 transgenic mice, and peptide-pulsed and ADC-1013-stimulated DCs induce antigen-specific T-cell proliferation in vitro. In vivo, treatment with ADC-1013 in a syngeneic bladder cancer model, negative for hCD40, induces significant antitumor effects and long-term tumor-specific immunity. Furthermore, ADC-1013 demonstrates significant antitumor effects in a human bladder cancer transplanted into immunodeficient NSG mice.
How is ADC 1013 administered?
Because ADC 1013 activates dendritic cells, it represents an immunomodulatory antibody for local immunotherapy of cancer. In the cancer environment, dendritic cells acquire many cancer antigens – boosting their activity and enhancing their expression of CD80 and CD86 leads to binding of CD28 on T-helper cells, which provokes a strong immune response. Stimulation of CD40 on dendritic cells initiates a process leading to a dramatic increase in specific T effector cells attacking the tumor. In addition, a tumor-specific memory is established leading to long term immunity to the cancer preventing growth and metastasis. The hope is that stimulation of dendritic cells with CD40 stimulation can overcome immune resistance that the cancer cells have induced against the cancer and its antigens.
A clinical study of intratumoral ADC 1013 has been initiated in patients with advanced solid tumors. It will be administered by intratumoral injection every second week for 8 weeks.
What does CD40 signaling mediate?
CD40, a TNFR (Tumor Necrosis Factor Receptor) family member, conveys signals regulating diverse cellular responses, ranging from proliferation and differentiation to growth suppression and cell death. First identified and functionally characterized on B-Cells, CD40 is expressed on a plethora of different cell types, including B-Cells, macrophages, dendritic cells, endothelial cells, and fibroblasts, and this widespread expression accounts for the central role of CD40 in the regulation of immune response and host defense (Ref.1). Binding of CD40 with its counter receptor, CD154 (also termedCD40L [CD40 ligand] or GP39), acts on Antigen presenting cells and T-Cells in a bi-directional fashion, mediating both humoral and cellular immune responses (Ref.2).
Unique to particular types of cells, CD40 engagement sets in motion a pattern of gene expression. B-Cells depend on CD40for survival, for expression of costimulatory molecules like B7 (to interact with T-Cells), germinal center formation, memory generation, Ig (Immunoglobulin) class switching and production of numerous cytokines and chemokines (IL-1, IL-6, IL-8,IL-10, IL-12 [Interleukins], TNF-Alpha [Tumor Necrosis Factor-Alpha], MIP1Alpha [Macrophage Inflammatory Protein-1Alpha] and cytotoxic radicals (Ref.3).
On the surface of dendritic cells, CD40–CD40L ligation regulates production of certain proinflammatory cytokines such as IL-8, MIP-1Alpha, TNF-Alpha and IL-12. Ligation of CD40 on monocytes is important in stimulating production of IL-1Alpha, IL-1Beta, TNF-Alpha, IL-6, and IL-8, as well as in the rescue of circulating monocytes from apoptotic death. Cell-to-cell contact via CD40–CD40L interactions is required for production of NO (Nitric Oxide) and IL-12by macrophages. NK cells induce B-Cell maturation, immunoglobulin secretion, and isotype switching—the pathways normally regulated by CD40–CD40L interactions. CD40 activates p44/42 MAPK through the Ras pathway in neuronal cells.CD40–CD40L interactions may also regulate proliferation and activation of effector cells such as mast cells, eosinophils, smooth muscle cells, and, most recently, human cultured myoblasts. Evidence of CD40 expression on non-immune cell types suggests a broader role of CD40 in cellular biology (Ref. 4).
CD40-mediated signal transduction induces the transcription of a large number of genes implicated in host defense against pathogens. This is accomplished by the activation of multiple pathways including NF-KappaB (Nuclear Factor-KappaB),MAPK (Mitogen-Activated Protein Kinase) and STAT3 (Signal Transducers and Activators of Transcription-3) (Ref.2) that regulate gene expression through activation of Activating Proteins, c-Jun, ATF2 (Activating Transcription Factor-2) and Rel transcription factors (Ref.3).
References
- CD40 induces apoptosis in carcinoma cells through activation of cytotoxic ligands of the tumor necrosis factor superfamily
- TTRAP, a novel protein that associates with CD40, tumor necrosis factor (TNF) receptor-75 and TNF receptor-associated factors (TRAFs), and that inhibits nuclear factor-kappa B activation
- Cooperation of multiple signaling pathways in CD40-regulated gene expression in B lymphocytes
- CD40 and CD154 in cell-mediated immunity



As you have displayed in the post, CD 40 antibody has realized great success. Maybe it will raise a revolution in modern antibody research.