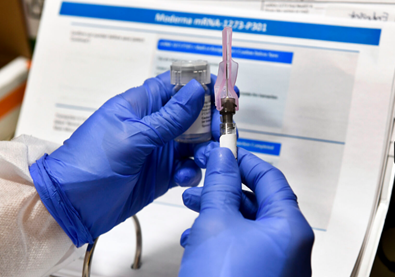
What We Know About Coronavirus Vaccine
By Garrett Belardi
National News Staff Writer
Today, Friday the 20th at 6:45 am both Pfizer Inc. and BioNTech SE announced a joint submission request to the U.S Drug and Food Administration for an Emergency Use Authorization of their vaccine in hopes to distribute their vaccines to high-risk locations by the middle to end of December 2020. Their joint submission rides on their 95% efficacy rate of the vaccines in the most recent round of testing. “Our work to deliver a safe and effective vaccine has never been more urgent, as we continue to see an alarming rise in the number of cases of COVID-19 globally. Filing in the U.S. represents a critical milestone in our journey to deliver a COVID-19 vaccine to the world and we now have a more complete picture of both the efficacy and safety profile of our vaccine, giving us confidence in its potential,” said Dr. Albert Bourla, Pfizer Chairman and CEO. If this submission is accepted by the FDA then the companies believe that it will instill confidence in the world and will hopefully begin to turn the tide against the coronavirus. Most drug-related testing takes on average 6-8 months to be confirmed by the FDA, but with the Emergency Use Authorization, the testing will be passed through and confirmed significantly faster than usual. “Pfizer and BioNTech’s combined manufacturing network has the potential to supply up to 50 million vaccine doses globally in 2020 and up to 1.3 billion doses by the end of 2021.” The two companies are confident with their ability to produce and distribute a significant amount of the vaccine in order to stave off the virus as fast as possible. However, Pfizer’s vaccine does not come without its own problems. Firstly, their vaccine BNT162b2 must be kept in specialized freezers at -70 degrees Celsius in order to allow it to survive. Secondly “anyone who takes it needs two doses, three weeks apart,” The Financial Review writes. This problem effectively cuts the total amount in half and also creates a delay of three weeks before the vaccine is allowed to fully function. Though the companies are still confident in their abilities to quickly work around these small issues they believe them to be saying, “Pfizer has vast experience and expertise in cold-chain shipping and has an established infrastructure to supply the vaccine worldwide, including distribution hubs that can store vaccine doses for up to six months.” The company also touted successfully delivering the vaccines to other countries without such machinery in past testing phases.
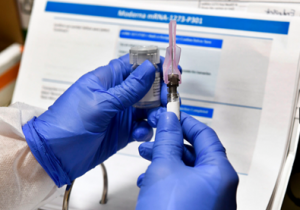
Overall, the news seems to be looking up for a potential vaccine for the virus for the end of the year. There is no telling if the FDA will confirm the two companies’ requests or not, and how long it might take until everyone is vaccinated. Nonetheless, there is reason to hope for a swift change in the tide of the coronavirus.
Contact Garrett at garrett.belardi@student.shu.edu